Abstract
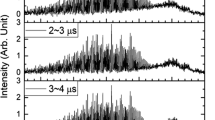
IN flash photolysis experiments involving methyl nitrite, transient absorption bands at 1999, 2036 and 2064 Å were seen1 faintly at 50 µsec, increasing in intensity slowly to a maximum at 2.5 msec and almost disappearing by 25 msec. These bands did not appear for any other nitrite tested. The reasons1 for these bands being attributed to CH3O· (being produced originally in a metastable excited state) were that this free radical, together with NO·, was expected to result from the alkyl nitrites which are in fact accepted sources of RO· (despite the early investigations2 in which the primary photochemical process in CH3ONO was deduced to be dissociation to the molecules HNO and H2CO), the observation of the expected partner fragment NO at the smallest delay times (50 µsec), and the virtual elimination of HNO, HCO, and formaldehyde in an excited state as the appropriate carrier.
Similar content being viewed by others
Article PDF
References
McGrath, W. D., and McGarvey, J. J., Nature, 201, 991 (1964).
Thompson, H. W., and Purkis, C. H., Trans. Faraday Soc., 32, 674, 1466 (1936).
Cottrell, T. L., The Strength of Chemical Bonds, second ed. (Butterworths, London, 1958).
Wijnen, M. H. J., J. Chem. Phys., 27, 710 (1957); 28, 271 (1958).
Black, G., and Porter, G., Proc. Roy. Soc., A, 266, 185 (1962).
Fujimoto, M., and Ingram, D. J. E., Trans. Farad. Soc., 30, 1304 (1958).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
OGILVIE, J. Electronic Absorption Spectrum of the CH3O-radical. Nature 208, 1315 (1965). https://doi.org/10.1038/2081315a0
Published:
Issue Date:
DOI: https://doi.org/10.1038/2081315a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.