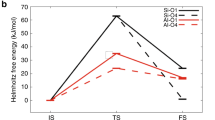
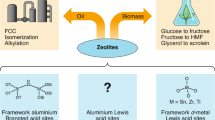
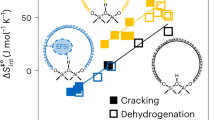
Abstract
SYNTHETIC zeolites1, especially those with the same structure as natural faujasite, have recently come into prominence as industrial catalysts. Pickert, Rabo, Dempsey, Schomaker and references2 have convincingly demonstrated the importance of the positions of the exchangeable cations which compensate for the effective negative charge of aluminium atoms in the aluminosilicate framework. ‘Linde’ 13X normally has Na+ in all the cation positions. Very little attention has been given to the effect of dehydration qf the zeolites on the position of the cations, or the position of individual ions when more than one type of cation is present.
Similar content being viewed by others
Article PDF
References
Breck, D. W., J. Chem. Educ., 41, 678 (1964). Broussard, L., and Shoemaker, D. P., J. Amer. Chem. Soc., 82, 1041 (1960).
Pickert, P. E., Rabo, J. A., Dempsey, E., and Schomaker, V., Proc. Third Intern. Cong. Catalysis, Amsterdam 1964 (North Holland Publishing Co., Amsterdam).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BARRY, T., LAY, L. Cation Sites in Synthetic Zeolites. Nature 208, 1312 (1965). https://doi.org/10.1038/2081312a0
Published:
Issue Date:
DOI: https://doi.org/10.1038/2081312a0
This article is cited by
-
EPR study of Mn2+ ions adsorption on silica gel
Czechoslovak Journal of Physics (1978)
-
Analysis of the EPR spectra of Mn2+ ions in synthetic zeolites at frequencies of 9,500 and 37,500 Mcps
Journal of Structural Chemistry (1967)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.