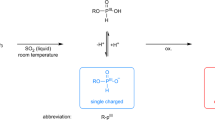
Abstract
PHENOLPHTHALEIN diphosphate was introduced by King1 as a substrate for phosphatases. Huggins and Talalay2 applied it to quantitative determinations of enzyme activities. They showed that the amount of phenolphthalein liberated by soluble phosphatases of animal origin increases more than linearly with the time of reaction. The same relation is found when varying the concentration of the enzyme instead of the time of reaction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
King, E. J., J. Path. and Bact., 55, 311 (1943).
Huggins, C., and Talalay, P., J. Biol. Chem., 159, 399 (1945).
Schmidt, G., Hecht, L., and Thannhauser, S., J. Biol. Chem., 178, 733 (1949).
Suomalainen, H., Linko, M., and Oura, E., Biochim. Biophys. Acta, 37, 482 (1960).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
PAUWELS, G. Occurrence of Phenolphthalein Monophosphate as an Intermediate in the Enzymatic Hydrolysis of Phenolphthalein Diphosphate to Phenolphthalein and Orthophosphate. Nature 202, 190–192 (1964). https://doi.org/10.1038/202190a0
Issue Date:
DOI: https://doi.org/10.1038/202190a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.