Abstract
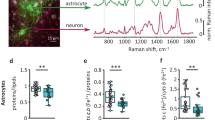
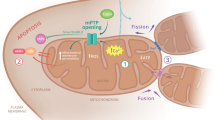
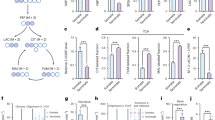
AN insoluble brain enzyme capable of oxidizing α-glyeerophosphate1 has been shown to be part of a purely mitochondrial system which resembles suc-cinate dehydrogenase in that pyridine nucleotides do not participate in electron transfer2. Histochemical localization of this activity in mammalian nervous tissue was made difficult by the fact that tetrazolium salts are ineffective electron-acceptors in preparations which oxidize α-glycerophosphate. Maximal reaction-rates, however, were obtained by using 2-methyl-1,4-naphthoquinone (menadione, K3) as intermediate acceptor. Histochemically, this effect was first demonstrated by Wattenberg in liver slices3. We found that menadione could be substituted by the sodium bisulphite derivative, but the diphosphoric acid ester (‘Synkavite’), vitamin K1, and 1,4-benzo-quinone were ineffective.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Green, D. E., Biochem. J., 30, 629 (1936).
Ringler, R. L., and Singer, T. P., Arch. Biophys. Biochem., 77, 229 (1958).
Wattenberg, L. W., and Leong, J. L., J. Histochem. Cytochem., 8, 296 (1960).
Slater, T. F., Nature, 183, 50 (1959).
Colpa-Boonstra, J. P., and Slater, E. C., Biochem. Biophys. Acta, 27, 122 (1958).
Sacktor, B., Packet, L., and Estabrook, R. W., Arch. Biophys Biochem., 80, 68 (1959).
Abood, L. G., and Alexander, L., J. Biol. Chem., 225, 717 (1957).
Giuditta, A., and Strecker, H. J., Biochem. Biophys. Res. Comm. 2, 159 (1960).
Wosilait, W., J. Biol. Chem., 235, 1196 (1960).
Hatefi, Y., Lester, R. L., Crane, F. L., and Widmer, C., Biochim. Biophys. Acta, 31, 490 (1959).
Redfearn, E. R., and Pumphrey, A. M., Biochem. J., 76, 64 (1960).
Abood, L. G., Brunngraber, E., and Taylor, M., J. Biol. Chem., 234, 1307 (1959).
Wosilait, W., and Nason, A., J. Biol. Chem., 206, 255 (1954).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
HESS, R., PEARSE, A. Histochemical and Homogenization Studies of Mitochondrial α-Glycerophosphate Dehydrogenase in the Nervous System. Nature 191, 718–719 (1961). https://doi.org/10.1038/191718a0
Issue Date:
DOI: https://doi.org/10.1038/191718a0
This article is cited by
-
Histochemical characteristics of a tonic smooth muscle
Histochemistry (1979)
-
On the subsarcolemmal localization of phenazine ethosulfate-linked ?-glycerophosphate dehydrogenase activity in pigeon pectoralis white muscle fibres
Histochemistry (1976)
-
The activity of mitochondrial α-glycerophosphate dehydrogenase in the rat nephron following triiodo-L-Thyronine treatment
Histochemistry (1976)
-
Morphogenesis of rat muscle spindles after nerve lesion during early postnatal development
Journal of Neurocytology (1976)
-
Histochemical basis of the correlation between RNP content and acticity of some oxidoreductases in the albino rat liver during experimental thyroid dysfunction
Bulletin of Experimental Biology and Medicine (1972)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.