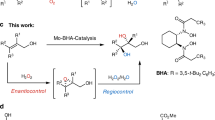
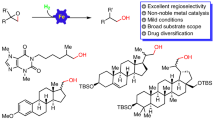
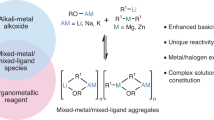
Abstract
INSPECTION of the literature reveals that hydrogenation of many, but not all, cyclic allylic alcohols gives products in which at least one hydrogen has been added to the same side of the molecule as the hydroxyl group. Thus, Schoenheimer and Evans1 reported that hydrogenation of allocholesterol (I) in amyl ether solution in the presence of a platinum catalyst gave coprostanol (in unstated yield). A re-investigation of this reaction was undertaken in an attempt to determine more precisely the influence of hydroxyl or other neighbouring groups on the stereochemistry of hydrogen addition.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Schoenheimer, R., and Evans, E. A., J. Biol. Chem., 114, 567 (1936).
Adams, R., and Shriner, R. L., J. Amer. Chem. Soc., 45, 2171 (1923).
Keenan, C. W., Giesemann, B. W., and Smith, H. A., J. Amer. Chem. Soc., 76, 229 (1954).
Wicker, R. J., J. Chem. Soc., 2165 (1956).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
DART, M., HENBEST, H. Effect of Alkali Salts on the Steric Course of Hydrogenation of a Cyclic Allylic Alcohol. Nature 183, 817–818 (1959). https://doi.org/10.1038/183817a0
Issue Date:
DOI: https://doi.org/10.1038/183817a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.