Abstract
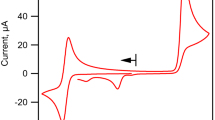
PYRIDINE 2-ALDOXIME forms well-defined coloured complexes with ferrous and with ferric ions in aqueous solution. The ferrous complex is violet-red in alkaline solution (with an absorption maximum at 530 mμ), reddish-orange in slightly acidic solution (with a maximum at 515 mμ), and golden-yellow below pH. 3 (with a maximum at 475 mμ). The ferric complex is reddish-brown in alkaline, neutral and slightly acidic solution (with no absorption maxima in the visible), and yellow in more acidic solution. All the colour changes are reversible.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Krause, R. A., and Busch, D. H., Nature, 181, 1529 (1958).
Whiteley, M. A., J. Chem. Soc., 24 (1903).
Brady, O. L., and Muers, M. M., J. Chem. Soc., 1599 (1930).
Brown, H. C., and Mihm, X. R., J. Amer. Chem. Soc., 77, 1723 (1955).
Charles, R. G., and Freiser, H., Anal. Chim. Acta, 11, 101 (1954).
Leussing, D. L., and Kolthoff, I. M., J. Amer. Chem. Soc., 75, 2476 (1953).
Milburn, R. M., J. Amer. Chem. Soc., 79, 537 (1957).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
HANANIA, G., IRVINE, D. Ferrous and Ferric Complexes with Pyridine 2-Aldoxime. Nature 183, 40–42 (1959). https://doi.org/10.1038/183040a0
Issue Date:
DOI: https://doi.org/10.1038/183040a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.