Abstract
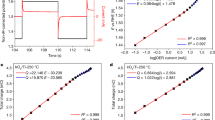
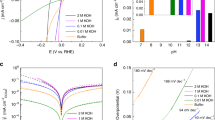
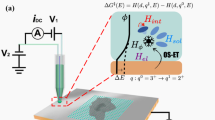
ELECTRON transfer reactions at the semiconductor – electrolyte interface are of particular interest because it is not known experimentally whether the reaction kinetics depend upon the bulk electron concentration in the semiconductor. Results of recent measurements of the hydrogen evolution reaction on single crystal germanium electrodes are presented here. The electrode potential (versus the normal hydrogen electrode) was measured under suitable conditions of purity for analysis of the mechanism (Table 1).
Similar content being viewed by others
Article PDF
References
Frumkin, Z. phys. Chem., 160, 116 (1932). Wagner and Traud, Z. Electrochem., 44, 391 (1938). Bockris, “Modern Aspects of Electrochemistry”, 253 (Butterworths Sci. Pub., 1954).
Bockris and Potter, J. Electrochem Soc., 99, 169 (1952).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
ERDÉLYI, V., GREEN, M. Hydrogen Overpotential on Germanium Electrodes. Nature 182, 1592 (1958). https://doi.org/10.1038/1821592b0
Issue Date:
DOI: https://doi.org/10.1038/1821592b0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.