Abstract
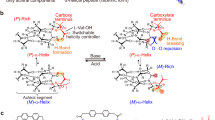
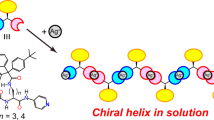
IN a communication from this laboratory1, the belief has been put forward that the polypeptide chains in poly-L-alanine fibres form right-handed rather than left-handed helices; this view is based on the X-ray diffraction pattern. If it is correct, it is to be expected that the same sense of helix will be the stable form in other L-polypeptides. Now the contribution to the optical rotation of an α-polypeptide from the helix alone is considerable; according to Fitts and Kirkwood2, a right-handed helix of polyglycine should have a specific rotation of about + 130°. In a later communication3 they have concluded from the change in specific rotation on destruction of the α-helix (in poly-γ-benzyl-L-glutamate and in poly-L-glutamic acid) that in these polymers the helices are right-handed.
Similar content being viewed by others
Article PDF
References
Elliott, A., and Malcolm, B. R., Nature [178, 912 (1956)].
Fitts, D. D., and Kirkwood, J. G., Proc. U.S. Nat. Acad. Sci., 42, 33 (1956).
Fitts, D. D., and Kirkwood, J. G., J. Amer. Chem. Soc., 78, 2650 (1956).
Doty, P., Holtzer, A. M., Bradbury, J. H., and Blout, E. R., J. Amer. Chem. Soc., 76, 4493 (1954). Doty, P., Bradbury, J. H., and Holtzer, A. M., ibid., 78, 947 (1956). Doty, P., and Yang, J. T., ibid., 78, 498 (1956).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
ELLIOTT, A., HANBY, W. & MALCOLM, B. Optical Rotation of the α-Helix in Synthetic Polypeptides. Nature 178, 1170 (1956). https://doi.org/10.1038/1781170a0
Issue Date:
DOI: https://doi.org/10.1038/1781170a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.