Abstract
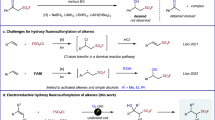
2,4-DINITROPHENYLHYDRAZONES have proved very useful derivatives for identification purposes, but have so far found only limited application in preparative work. This is due to the fact that there exists no general method for the regeneration of the corresponding carbonyl compounds. As Djerassi pointed out1, the failure to cleave 2,4-dinitrophenyl-hydrazones has imposed considerable limitations on the use of the Mattox–Kendall2 dehydrobromination method in the field of sterol chemistry. Thus, in the case of 2,4-dinitrophenylhydrazones of Δ4,6-dien-3-ketosteroids, only about 5–10 per cent of ketone could be recovered after treatment with pyruvic acid and hydrobromic acid, and derivatives of Δ1,4-dien-3-ketosteroids could not be split at all1.
Similar content being viewed by others
Article PDF
References
Djerassi, C., J. Amer. Chem. Soc., 71, 1003 (1949).
Mattox, V. R., and Kendall, E. C., J. Amer. Chem. Soc., 70, 882 (1948); 72, 2290 (1950).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
DEMAECKER, J., MARTIN, R. A Method for the Cleavage of 2,4-Dinitrophenylhydrazones and its Application to Steroidal Ketones. Nature 173, 266 (1954). https://doi.org/10.1038/173266a0
Issue Date:
DOI: https://doi.org/10.1038/173266a0
This article is cited by
-
Total synthesis ofdl-9(11)-dehydrotestosterone anddl-testosterone
Proceedings / Indian Academy of Sciences (1978)
-
Regeneration of carbonyl compounds from 2,4‐dinitrophenylhydrazones with sulfuric acid
Journal of the American Oil Chemists' Society (1960)
-
Pathway of Carbon-14 Incorporation from NaH14CO3 to Hæm in Duck Blood incubated in vitro
Nature (1959)
-
Cleavage of the 2 : 4-Dinitrophenylhydrazones of Ketones
Nature (1954)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.