Abstract
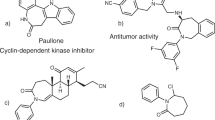
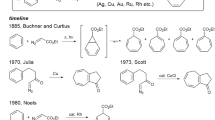
ALTHOUGH several syntheses of azulene (VIII; R=H) and azulene derivatives have deen described1, investigations on the chemistry of this non-benzenoid aromatic system have been hampered by the fact that the procedures hitherto available do not readily lend themselves to the preparation of azulenes on more than a relatively small scale. We have recently developed a new synthesis, based on the method of alkenylation with lithium alkenyls2, which offers some advantages in this respect. 
Similar content being viewed by others
Article PDF
References
For references, cf. Pommer, Angew. Chem., 62, 281 (1950).
Braude, Coles and Timmons, Nature, 166, 58 (1950); J. Chem. Soc., 2000, 2007, 2012, 2014 (1950); 2078 (1951). Braude and Forbes, J. Chem. Soc., 1755 (1951).
Favorskii and Domnin, J. Gen. Chem. U.S.S.R., 6, 727 (1936).
Braude and Forbes, J. Chem. Soc. (forthcoming paper).
Ball, Goodwin and Morton, Biochem. J., 42, 516 (1948); Wendler, Slates and Tishler, J. Amer. Chem. Soc., 71, 3267 (1949).
Nazarov et al., Bull. Acad. Sci. U.S.S.R., Cl. Sci. Chim., 633 (1946); 205 (1947); J. Gen. Chem. U.S.S.R., 20, 1441 (1950).
Plattner and Büchi, Helv. Chim. Acta, 29, 1608 (1946).
Cook, Philip and Somerville, J. Chem. Soc., 164 (1948).
Anderson and Nelson, J. Amer. Chem. Soc., 73, 232 (1951).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BRAUDE, E., FORBES, W. A New Route to Azulenes. Nature 168, 874 (1951). https://doi.org/10.1038/168874a0
Issue Date:
DOI: https://doi.org/10.1038/168874a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.