Abstract
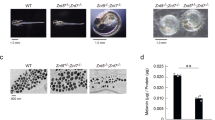
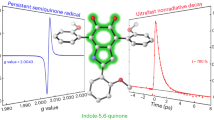
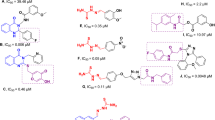
SOME years ago it was suggested that the first step in melanogenesis, the oxidation of tyrosine to o-dihydroxyphenylalanine, can be represented by the following general reactions: (1) o-dihydroxyphenol → o-quinone → intermediate products → melanin; (2) o-quinone + monohydroxyphenol + H2O = 2 o-dihydroxyphenol; and that of these reactions only the first is really enzymic1.
Similar content being viewed by others
Article PDF
References
Califano, L., and Kertész, D., Nature, 142, 1036 (1938); Enzymologia, 6, 233 (1939).
Kertész, D., Bull. Soc. Chirm. Biol., 32, 587 (1950); ibid. (in the press).
Lerner, A. B., Fitzpatrick, T. B., Calkins, E., and Summerson, W. H., J. Biol. Chem., 178, 185 (1949).
Kubowitz, F., Biochem. Z., 298, 32 (1938).
Keilin, D., and Mann, T., Proc. Roy. Soc., B, 125, 187 (1938).
Abbott, L. D., and James, G. W., J. Lab. Clin. Med., 35, 35 (1950).
Harley-Mason, J., and Bu'Lock, J. D., Nature, 166, 1036 (1950).
Mallette, M. F., and Dawson, C. R., Arch. Biochem., 23, 29 (1949).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
KERTÉSZ, D. Tyrosinase and Polyphenoloxidase; the Role of Metallic Ions in Melanogenesis. Nature 168, 697 (1951). https://doi.org/10.1038/168697a0
Issue Date:
DOI: https://doi.org/10.1038/168697a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.