Abstract
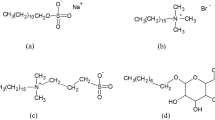
IT is well known that anionic and cationic detergents are not compatible in aqueous phase when they are present in equimolecular amounts. Compatibility is only observed for certain ratios which depend upon the molecular structure of both components. Precipitation of anionic by cationic detergents present in equimolar amounts constitutes the basis of one of the methods used for their estimation. The work carried out by Salton and Alexander1 has shown this method to be applicable for determining small amounts of such pure detergents.
Similar content being viewed by others
Article PDF
References
Salton, M. R. J., and Alexander, A. E., Research, 2, 247 (1949).
Schulman, J. H., Trans. Farad. Soc., 33, 1116 (1937).
Matalon, R. (unpublished work)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
MATALON, R., SALTON, M. & COHEN, M. Molecular Interaction and its Relation to the Formation of Sodium Dodecylsulphate–Cetyltrimethylammonium Bromide Complex. Nature 167, 241 (1951). https://doi.org/10.1038/167241a0
Issue Date:
DOI: https://doi.org/10.1038/167241a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.