Abstract
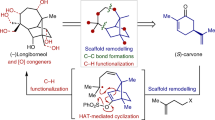
THE configuration of the C/D ring fusion in equilenin has been the subject of a number of communications1–3. We consider that the arguments advanced for a trans ring junction leave much to be desired. Shoppee's1 conclusion that the dehydrogenation of equilin to equilenin under mild conditions proves the steric identity of the C/D union in these two substances can no longer be accepted, since isoequilin, which is now known4,5 to be also trans, gives isoequilenin (which has the opposite C/D configuration to equilenin) under the same conditions6.
Similar content being viewed by others
Article PDF
References
Shoppee, Nature, 161, 207 (1948).
Rosenkranz, Djerassi, Kaufmann, Pataki and Romo, Nature, p. 814 of this issue.
Klyne, Nature, 161, 434 (1948).
Heer and Miescher, Helv. Chim. Acta, 31, 1289 (1948).
Singh, G., J. Amer. Chem. Soc. (in the press).
Hirschmann, H., and Wintersteiner, J. Biol. Chem., 126, 737 (1938).
Barton, J. Chem. Soc., 783 (1948).
Bachmann, Cole and Wilds, J . Amer. Chem. Soc., 62, 824 (1940).
Dane and Schmitt, Ann., 537, 246 (1939).
See, for example, Anner and Miescher, Helv. Chim. Acta, 30, 1422 (1947).
Pearlman and Wintersteiner, J. Biol. Chem., 130, 35 (1939); 132, 605 (1940).
Inhoffen and Zühlsdorff, Ber., 74, 1911 (1941).
Wilds and Djerassi, J. Amer. Chem. Soc., 68, 2125 (1946).
Wieland and Dane, Z. Physiol. Chem., 216, 91 (1933).
Peak, Nature, 140, 280 (1937).
Dimroth and Jonsson, Ber., 74, 5201 (1941).
Windaus and Zühlsdorff, Ann., 536, 209 (1938).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
STORK, G., SINGH, G. Configuration of the C/D Ring Junction in Equilenin and Other Steroids. Nature 165, 816 (1950). https://doi.org/10.1038/165816a0
Issue Date:
DOI: https://doi.org/10.1038/165816a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.