Abstract
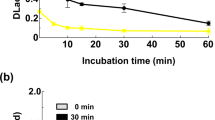
IT was shown that laccase, which had been inactivated by cyanide under certain conditions, could be almost completely re-activated by cupric ions, whereas other metals were ineffective1. Further evidence will be presented here to show that copper is an essential constituent of this enzyme. Laccase was purified by the method of D. Bertrand2,3. At each successive stage of purification the enzyme activity was measured in the manner previously described1, and copper and manganese were estimated by microchemical methods4,5. Manganese disappears after the first steps of purification; the copper content increases with the and remains proportional to activity in all fractions up to the purest preparation having 30,000 in presence of paraphenylene diamine (Fig. 1).
Similar content being viewed by others
Article PDF
References
Tissières, A., Nature, 162, 340 (1948).
Bertrand, D., Bull. Soc. Chim. biol., 26, 40 (1944).
Bertrand, D., Ann. Inst. Pasteur, 73, 266 (1947).
Eden, A., and Green, H. H., Biochem. J., 34, 1203 (1940).
Wiese, A. C., and Johnson, B. C., J. Biol. Chem., 127, 203 (1939).
Bertrand, D., Bull. Soc. Chim. biol., 26, 45 (1944).
Bertrand, D., Belval, H., and Legrand, Gilberte, C.R. Acad. Sci., Paris, 223, 1189 (1946).
Bertrand, D., Belval, H., and Legrand, Gilberte, Bull. Soc. Chim. biol., 29, 607 (1947).
Bertrand, D., Bull. Soc. Chim. biol., 29, 608 (1947).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
TISSIÈRES, A. Constitution and Properties of Laccase. Nature 163, 480 (1949). https://doi.org/10.1038/163480a0
Issue Date:
DOI: https://doi.org/10.1038/163480a0
This article is cited by
-
Copper formate–lysine nanoparticles with polyphenol oxidase-like activity for the detection of epinephrine
Analytical and Bioanalytical Chemistry (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.