Abstract
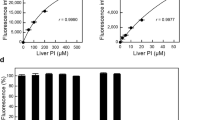
IT is generally assumed that when phosphocreatine is hydrolysed either enzymatically or spontaneously, the end products are creatine and phosphoric acid. This, however, is not always found to be the case. When phosphocreatine is hydrolysed at 38° in saturated picric acid, about 10 percent of the substance is converted into creatinine (Fig. 1.). Under these conditions, creatine is not transformed into creatinine. Obviously phosphocreatine has a certain spontaneous tendency to ring formation. If heated to 100° either in picric acid or hydrochloric acid, there is a rapid formation of creatinine, followed by a linear increase due to the reaction creatine - creatinine (Fig. 2.). If the two curves are subtracted, a curve of much the same appearance as Fig. 1 results. Whether picric or hydrochloric acid is used for hydrolysis is of no importance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Lundquist, F., Acta Pharm. et Tox., 1, 307 (1945).
Rehberg, P. B., XVI Int. Physiol. Kongress, Kongressber., 1, 4 (1938).
Abdon, N-O., Kungl. Fysiograf. Sälsk. i Lund Förhandl., 5, 14 (1935).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
LUNDQUIST, F. Splitting of Phosphocreatine. Nature 159, 98–99 (1947). https://doi.org/10.1038/159098a0
Issue Date:
DOI: https://doi.org/10.1038/159098a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.