Abstract
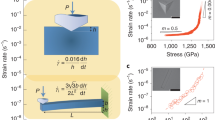
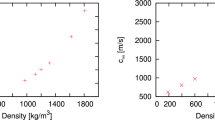
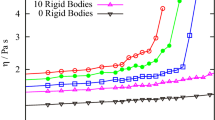
THE viscosity, η, of a fluid can be represented by1 where N is Avogadro's number; c is a packing factor; m is the mass of a molecule; k is Boltzmann's constant; Evisc. is the energy of activation of viscous flow; V is the molecular volume; ET is the sum of the potential and the kinetic energy of the molecules. We have found that ET can be expressed as the sum of the latent heat of vaporization (ΔEvap.) and a multiple of R.T (d.R.T). It has been shown2 that Evisc. equals the work of cohesion for non-associated liquids, and from this fact it was deduced that Evisc. equals the bond energy between two interacting molecules. Further, it has since been deduced that ΔEvap. = b.Evisc., where b is a factor depending on the mean co-ordination. Thus both ΔEvap. and Evisc. depend on the number of bonds per mole, formed by the interaction of the molecules.
Similar content being viewed by others
Article PDF
References
Guareschi, P., Atti Acad. Sci. Torino, Disp. 2a, 118 (1939).
Grunberg, L., and Nissan, A. H., Nature, 154, 146 (1944).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
GRUNBERG, L., NISSAN, A. Viscosity of Ordinary Liquids at High Rates of Shear. Nature 156, 241 (1945). https://doi.org/10.1038/156241a0
Issue Date:
DOI: https://doi.org/10.1038/156241a0
This article is cited by
-
Rheologische VorgÄnge als ein mechanisch-thermodynamisches Problem
Kolloid-Zeitschrift (1953)
-
Viskosität und Konstitution
Kolloid-Zeitschrift (1952)
-
Temporary Reduction of Viscosity of Liquids at High Rates of Shear
Nature (1951)
-
Viscosity of Liquids at High Rates of Shear
Nature (1950)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.