Abstract
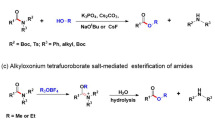
ALTHOUGH, as pointed out in a recent review1, acid or alkali catalysed hydrolysis or esterification and, it may be added, alkoxy interchange, usually take place by mechanisms in which the bonds of the alkoxyl carbon atom are not disturbed (mechanism I), the alternative mechanism (II) does, however, occur, and probably to a greater extent than has previously been recognized, when the alkoxyl group R" has electron–releasing properties.
Similar content being viewed by others
Article PDF
References
Watson, Ann. Repts. Progress of Chemistry, 27, 229 (1940).
Arcus and Kenyon, J. Chem. Soc., 1912 (1938).
Kenyon, Partridge and Phillips, J. Chem. Soc., 216 (1937).
Reference (2) and unpublished results.
"Organic Syntheses", 6, 68.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BALFE, M., KENYON, J. Reactions of Carboxylic Esters. Nature 148, 196 (1941). https://doi.org/10.1038/148196a0
Issue Date:
DOI: https://doi.org/10.1038/148196a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.