Abstract
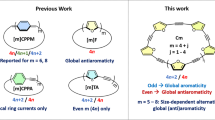
SOME time ago, I completed an X-ray analysis of the crystal structure of dibenzyl1, and showed that the results could best be explained by a three-dimensional model of the molecule, in which the planes of the benzene rings were at right angles to the plane containing the zig-zag of the connecting CH2 groups, as shown in Fig. 1. The structure is thus an interesting contrast to those aromatic compounds like naphthalene and durene in which all the carbon atoms of the molecule are found to lie in one plane. FIG. 1.
Similar content being viewed by others
Article PDF
References
Proc. Roy. Soc, A, 1934, in the press.
Current Science, 2, 480, June 1934.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
ROBERTSON, J. Shape of the Dibenzyl Molecule. Nature 134, 381 (1934). https://doi.org/10.1038/134381a0
Issue Date:
DOI: https://doi.org/10.1038/134381a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.