Abstract
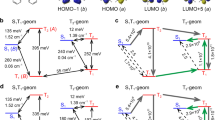
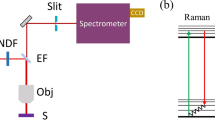
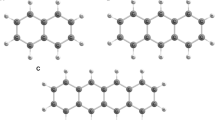
RECENT work on the Raman effect has indicated that the frequencies so determined may be attributed to vibrational frequencies, or combinations of such frequencies, of the normal electronic state of the molecule. It is therefore to be expected that these levels will appear as end states in the process of emission or fluorescence. This is neatly confirmed in the case of benzol.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
SHAPIRO, C. Raman Spectrum and Fluorescence of Benzol. Nature 124, 372 (1929). https://doi.org/10.1038/124372b0
Issue Date:
DOI: https://doi.org/10.1038/124372b0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.