Abstract
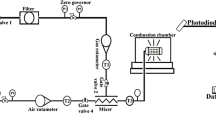
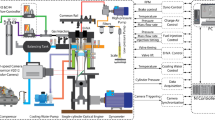
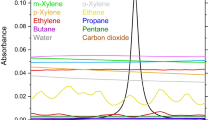
MR. HUGHES'S letter raises some interesting questions regarding the conditions under which the “blue flame” of the coal fire appears. There is no reason to suppose that under appropriate conditions the flame of burning carbon monoxide cannot be seen in a coal fire, but it would be difficult to identify since the spectrum is, in the main, continuous. On the other hand, the blue flame of copper chloride, which is distinct from the green flame of the oxide, has a very characteristic spectrum, and there is no difficulty in its identification. There is no doubt, however, that for the appearance of the spectrum of a compound certain accessory conditions have to be fulfilled, and in many cases their effect is by no means obvious. Perhaps one of the most striking instances of this is to be found in the appearance of the spectrum associated with burning sulphur in the flame of an ordinary bunsen burner when the gases of the flame are cooled, e.g. by holding a thick plate of cold metal in the flame. In this case the sulphur occurs as an impurity in the coal gas but is not seen in the spectrum of the burning gases unless they are cooled.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
MERTON, T. [Letters to Editor]. Nature 109, 683 (1922). https://doi.org/10.1038/109683b0
Issue Date:
DOI: https://doi.org/10.1038/109683b0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.