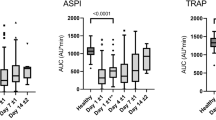
Abstract
Background
In the Fontan palliation for single ventricle heart disease (SVHD), pulmonary blood flow is non-pulsatile/passive, low velocity, and low shear, making viscous power loss a critical determinant of cardiac output. The rheologic properties of blood in SVHD patients are essential for understanding and modulating their limited cardiac output and they have not been systematically studied. We hypothesize that viscosity is decreased in single ventricle circulation.
Methods
We evaluated whole blood viscosity, red blood cell (RBC) aggregation, and RBC deformability to evaluate changes in healthy children and SVHD patients. We altered suspending media to understand cellular and plasma differences contributing to rheologic differences.
Results
Whole blood viscosity was similar between SVHD and healthy at their native hematocrits, while viscosity was lower at equivalent hematocrits for SVHD patients. RBC deformability is increased, and RBC aggregation is decreased in SVHD patients. Suspending SVHD RBCs in healthy plasma resulted in increased RBC aggregation and suspending healthy RBCs in SVHD plasma resulted in lower RBC aggregation.
Conclusions
Hematocrit corrected blood viscosity is lower in SVHD vs. healthy due to decreased RBC aggregation and higher RBC deformability, a viscous adaptation of blood in patients whose cardiac output is dependent on minimizing viscous power loss.
Impact
-
Patients with single ventricle circulation have decreased red blood cell aggregation and increased red blood cell deformability, both of which result in a decrease in blood viscosity across a large shear rate range.
-
Since the unique Fontan circulation has very low-shear and low velocity flow in the pulmonary arteries, blood viscosity plays an increased role in vascular resistance, therefore this work is the first to describe a novel mechanism to target pulmonary vascular resistance as a modifiable risk factor.
-
This is a novel, modifiable risk factor in this patient population.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Fontan, F. & Baudet, E. Surgical repair of tricuspid atresia. Thorax 26, 240–248 (1971).
Pundi, K. N. et al. 40-year follow-up after the Fontan operation: long-term outcomes of 1,052 patients. J. Am. Coll. Cardiol. 66, 1700–1710 (2015).
d’Udekem, Y. et al. The Fontan procedure: contemporary techniques have improved long-term outcomes. Circulation 116, I157–I164 (2007).
Mitchell, M. E. et al. Intermediate outcomes after the Fontan procedure in the current era. J. Thorac. Cardiovasc Surg. 131, 172–180 (2006).
Dabal, R. J. et al. The modern Fontan operation shows no increase in mortality out to 20 years: a new paradigm. J. Thorac. Cardiovasc. Surg. 148, 2517–2523.e2511 (2014).
Perrin, N. et al. The Fontan circulation: from ideal to failing hemodynamics and drug therapies for optimization. Can. J. Cardiol. 38, 1059–1071 (2022).
Khairy, P. et al. Long-term survival, modes of death, and predictors of mortality in patients with Fontan surgery. Circulation 117, 85–92 (2008).
Weinreb, S. J. et al. End-organ Function and exercise performance in patients with Fontan circulation: what characterizes the high performers? J. Am. Heart Assoc. 9, e016850 (2020).
Paridon, S. M. et al. A cross-sectional study of exercise performance during the first 2 decades of life after the Fontan operation. J. Am. Coll. Cardiol. 52, 99–107 (2008).
Gewillig, M. & Goldberg, D. J. Failure of the Fontan circulation. Heart Fail Clin. 10, 105–116 (2014).
Gewillig, M. & Brown, S. C. The Fontan circulation after 45 years: update in physiology. Heart 102, 1081–1086 (2016).
Cheng, A. L. et al. Elevated low-shear blood viscosity is associated with decreased pulmonary blood flow in children with univentricular heart defects. Pediatr. Cardiol. 37, 789–801 (2016).
Jeremiasen, I., Tran-Lundmark, K., Idris, N., Tran, P. K. & Moledina, S. Pulmonary vasodilator therapy in children with single ventricle physiology: effects on saturation and pulmonary arterial pressure. Pediatr. Cardiol. 41, 1651–1659 (2020).
Park, I. S. Efficacy of pulmonary vasodilator therapy in patients with functionally single ventricle. Int. Heart J. 56, S26–S30 (2015).
Snarr, B. S., Paridon, S. M., Rychik, J. & Goldberg, D. J. Pulmonary vasodilator therapy in the failing Fontan circulation: rationale and efficacy. Cardiol. Young. 25, 1489–1492 (2015).
Agnoletti, G. et al. Endothelin inhibitors lower pulmonary vascular resistance and improve functional capacity in patients with Fontan circulation. J. Thorac. Cardiovasc. Surg. 153, 1468–1475 (2017).
Goldberg, D. J. et al. Impact of oral sildenafil on exercise performance in children and young adults after the Fontan operation: a randomized, double-blind, placebo-controlled, crossover trial. Circulation 123, 1185–1193 (2011).
Schuuring, M. J. et al. Impact of Bosentan on exercise capacity in adults after the Fontan procedure: a randomized controlled trial. Eur. J. Heart Fail 15, 690–698 (2013).
Li, D., Zhou, X., An, Q. & Feng, Y. Pulmonary vasodilator therapy after the Fontan procedure: a meta-analysis. Heart Fail Rev. 26, 91–100 (2021).
Latus, H. et al. Evaluation of pulmonary endothelial function in Fontan patients. J. Thorac. Cardiovasc. Surg. 158, 523–531.e521 (2019).
Henaine, R. et al. Effects of lack of pulsatility on pulmonary endothelial function in the Fontan circulation. J. Thorac. Cardiovasc. Surg. 146, 522–529 (2013).
Kurotobi, S. et al. Bidirectional cavopulmonary shunt with right ventricular outflow patency: the impact of pulsatility on pulmonary endothelial function. J. Thorac. Cardiovasc. Surg. 121, 1161–1168 (2001).
Khambadkone, S. et al. Basal pulmonary vascular resistance and nitric oxide responsiveness late after Fontan-type operation. Circulation 107, 3204–3208 (2003).
Whitehead, K. K. et al. Nonlinear power loss during exercise in single-ventricle patients after the Fontan: insights from computational fluid dynamics. Circulation 116, I165–I171 (2007).
Deshaies, C. et al. Thromboembolic risk after atriopulmonary, lateral tunnel, and extracardiac conduit Fontan surgery. J. Am. Coll. Cardiol. 74, 1071–1081 (2019).
Kelly, J. M. et al. Evaluating the longevity of the Fontan pathway. Pediatr. Cardiol. 41, 1539–1547 (2020).
Deal, B. J. & Jacobs, M. L. Management of the failing Fontan circulation. Heart 98, 1098–1104 (2012).
Mahle, W. T., Todd, K. & Fyfe, D. A. Endothelial function following the Fontan operation. Am. J. Cardiol. 91, 1286–1288 (2003).
Jin, S. M., Noh, C. I., Bae, E. J., Choi, J. Y. & Yun, Y. S. Impaired vascular function in patients with Fontan circulation. Int J. Cardiol. 120, 221–226 (2007).
Inai, K., Saita, Y., Takeda, S., Nakazawa, M. & Kimura, H. Skeletal muscle hemodynamics and endothelial function in patients after Fontan operation. Am. J. Cardiol. 93, 792–797 (2004).
Natarajan, S. et al. Peripheral arterial function in infants and young children with one-ventricle physiology and hypoxemia. Am. J. Cardiol. 103, 862–866 (2009).
Wei, Z. A. et al. The advantages of viscous dissipation rate over simplified power loss as a Fontan hemodynamic metric. Ann. Biomed. Eng. 46, 404–416 (2018).
Tomkiewicz-Pajak, L. et al. Iron deficiency and hematological changes in adult patients after Fontan operation. J. Cardiol. 64, 384–389 (2014).
Härtel, J. A. et al. Altered hemorheology in Fontan patients in normoxia and after acute hypoxic exercise. Front. Physiol. 10, 1443 (2019).
Waltz, X. et al. Is there a relationship between the hematocrit-to-viscosity ratio and microvascular oxygenation in brain and muscle? Clin. Hemorheol. Microcirc. 59, 37–43 (2015).
Fan, F. C., Chen, R. Y., Schuessler, G. B. & Chien, S. Effects of hematocrit variations on regional hemodynamics and oxygen transport in the dog. Am. J. Physiol. 238, H545–522 (1980).
Nogueira, J. R. Clinical evolution of hemorrhage in the proximal portion of the digestive tract. Comparison of its management with and without endoscopy. Rev. Gastroenterol. Mex. 42, 9–17 (1977).
Baer, R. W., Vlahakes, G. J., Uhlig, P. N. & Hoffman, J. I. Maximum myocardial oxygen transport during anemia and polycythemia in dogs. Am. J. Physiol. 252, H1086–H1095 (1987).
Gagnon, D. R., Zhang, T. J., Brand, F. N. & Kannel, W. B. Hematocrit and the risk of cardiovascular disease—the Framingham study: a 34-year follow-up. Am. Heart J. 127, 674–682 (1994).
Restrepo, M. et al. Energetic implications of vessel growth and flow changes over time in Fontan patients. Ann. Thorac. Surg. 99, 163–170 (2015).
Low, H. T., Chew, Y. T. & Lee, C. N. Flow studies on atriopulmonary and cavopulmonary connections of the Fontan operations for congenital heart defects. J. Biomed. Eng. 15, 303–307 (1993).
de Leval, M. R., Kilner, P., Gewillig, M. & Bull, C. Total cavopulmonary connection: a logical alternative to atriopulmonary connection for complex Fontan operations. Experimental studies and early clinical experience. J. Thorac. Cardiovasc. Surg. 96, 682–695 (1988).
Dubini, G., de Leval, M. R., Pietrabissa, R., Montevecchi, F. M. & Fumero, R. A numerical fluid mechanical study of repaired congenital heart defects. Application to the total cavopulmonary connection. J. Biomech. 29, 111–121 (1996).
de Leval, M. R. et al. Use of computational fluid dynamics in the design of surgical procedures: application to the study of competitive flows in cavo-pulmonary connections. J. Thorac. Cardiovasc. Surg. 111, 502–513 (1996).
Ensley, A. E. et al. Fluid mechanic assessment of the total cavopulmonary connection using magnetic resonance phase velocity mapping and digital particle image velocimetry. Ann. Biomed. Eng. 28, 1172–1183 (2000).
Healy, T. M., Lucas, C. & Yoganathan, A. P. Noninvasive fluid dynamic power loss assessments for total cavopulmonary connections using the viscous dissipation function: a feasibility study. J. Biomech. Eng. 123, 317–324 (2001).
Wei, Z. et al. Non-Newtonian effects on patient-specific modeling of Fontan hemodynamics. Ann. Biomed. Eng. 48, 2204–2217 (2020).
Cheng, A. L., Pahlevan, N. M., Rinderknecht, D. G., Wood, J. C. & Gharib, M. Experimental investigation of the effect of non-Newtonian behavior of blood flow in the Fontan circulation. Eur. J. Mech. B Fluids 68, 184–192 (2018).
Kovacs, G., Olschewski, A., Berghold, A. & Olschewski, H. Pulmonary vascular resistances during exercise in normal subjects: a systematic review. Eur. Respir. J. 39, 319–328 (2012).
Goldberg, D. J. et al. Results of the fuel trial. Circulation 141, 641–651 (2020).
Gewillig, M. et al. Volume load paradox while preparing for the fontan: not too much for the ventricle, not too little for the lungs. Interact. Cardiovasc Thorac. Surg. 10, 262–265 (2010).
Schmid-Schönbein, H., Wells, R. & Goldstone, J. Influence of deformability of human red cells upon blood viscosity. Circ. Res. 25, 131–143 (1969).
Alexy, T. et al. Physical properties of blood and their relationship to clinical conditions. Front. Physiol. 13, 906768 (2022).
Schmid-Schönbein, H., Gaehtgens, P. & Hirsch, H. On the shear rate dependence of red cell aggregation in vitro. J. Clin. Invest 47, 1447–1454 (1968).
Armstrong, J. K., Wenby, R. B., Meiselman, H. J. & Fisher, T. C. The Hydrodynamic Radii of macromolecules and their effect on red blood cell aggregation. Biophys. J. 87, 4259–4270 (2004).
Linderkamp, O., Ozanne, P., Wu, P. Y. & Meiselman, H. J. Red blood cell aggregation in preterm and term neonates and adults. Pediatr. Res. 18, 1356–1360 (1984).
Long, J. A., Undar, A., Manning, K. B. & Deutsch, S. Viscoelasticity of pediatric blood and its implications for the testing of a pulsatile pediatric blood pump. ASAIO J. 51, 563–566 (2005).
Acknowledgements
We would like to acknowledge the clinical teams in the cardiac catheterization laboratory and the magnetic resonance imaging center at Children’s Hospital Los Angeles, without these incredible teams this research could not have happened.
Funding
This work is supported by funding from National Heart, Lung and Blood Institute, NIH# 1 K23 HL 119627–01A1 (J.A.D.), NIH# 1 U01 HL 117718–01 (J.A.D., T.D.C., J.C.W.), and Additional Ventures Foundation (J.A.D., J.C.W.).
Author information
Authors and Affiliations
Contributions
J.A.D. and S.S. designed the study. S.S., H.L., R.W., A.L.C. and J.A.D. conducted the experiments and acquired data. S.S., J.A.D., J.C.W., and H.J.M. analyzed data. All authors were involved in writing the manuscript. All authors reviewed the results and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
JAD served on the scientific advisory board for Alcor Scientific in 2022 but this has no relation to the research presented in this article.
Consent for publication
The Institutional Review Board at Children’s Hospital Los Angeles reviewed and approved the research protocol. Consent and/or Assent was required from all parents and/or participants in this research study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Suriany, S., Liu, H., Cheng, A.L. et al. Decreased erythrocyte aggregation in Glenn and Fontan: univentricular circulation as a rheologic disease model. Pediatr Res 95, 1335–1345 (2024). https://doi.org/10.1038/s41390-023-02969-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-023-02969-5